|
This means that sulfur can share ONE electron with EACH of two fluorine atoms, completing all of their octets simultaneously. 
Sulfur brings 6 valence electrons with it, and so needs two extra for have a full octet.įluorine brings 7 valence electrons with it, and so needs one extra to complete its octet. They are both non-metals, so they share electrons to form covalent bonds. 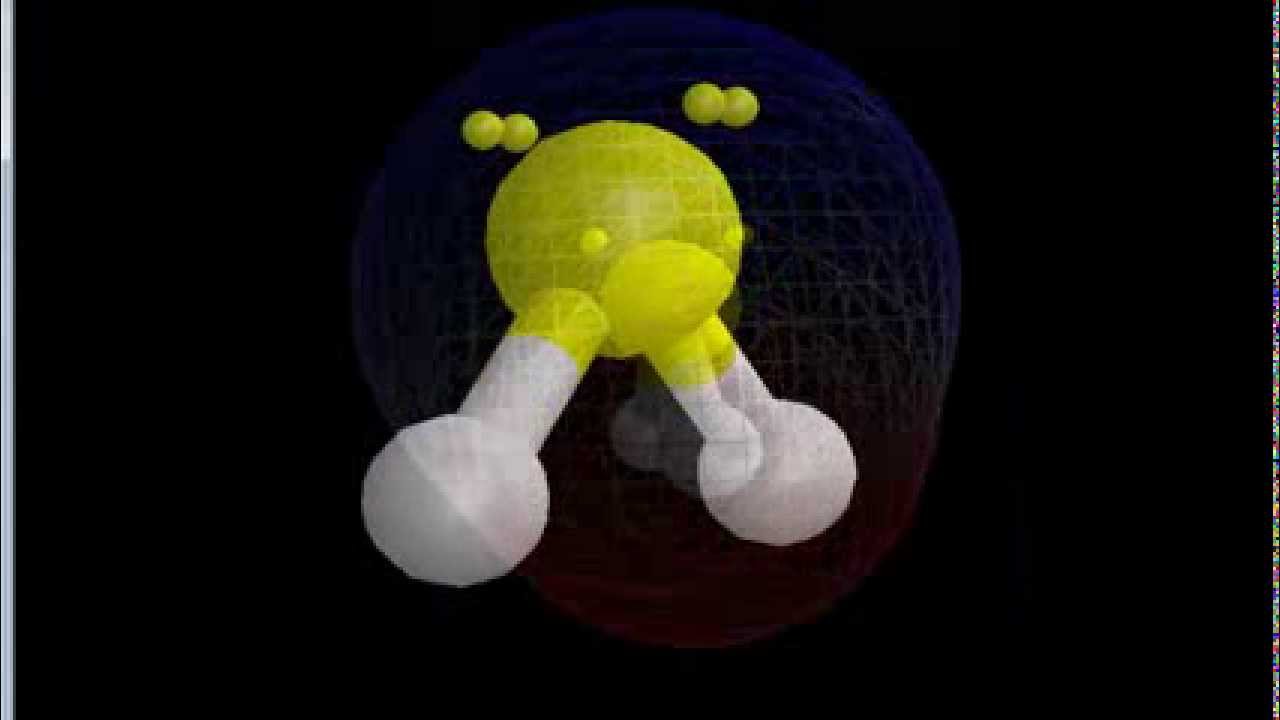 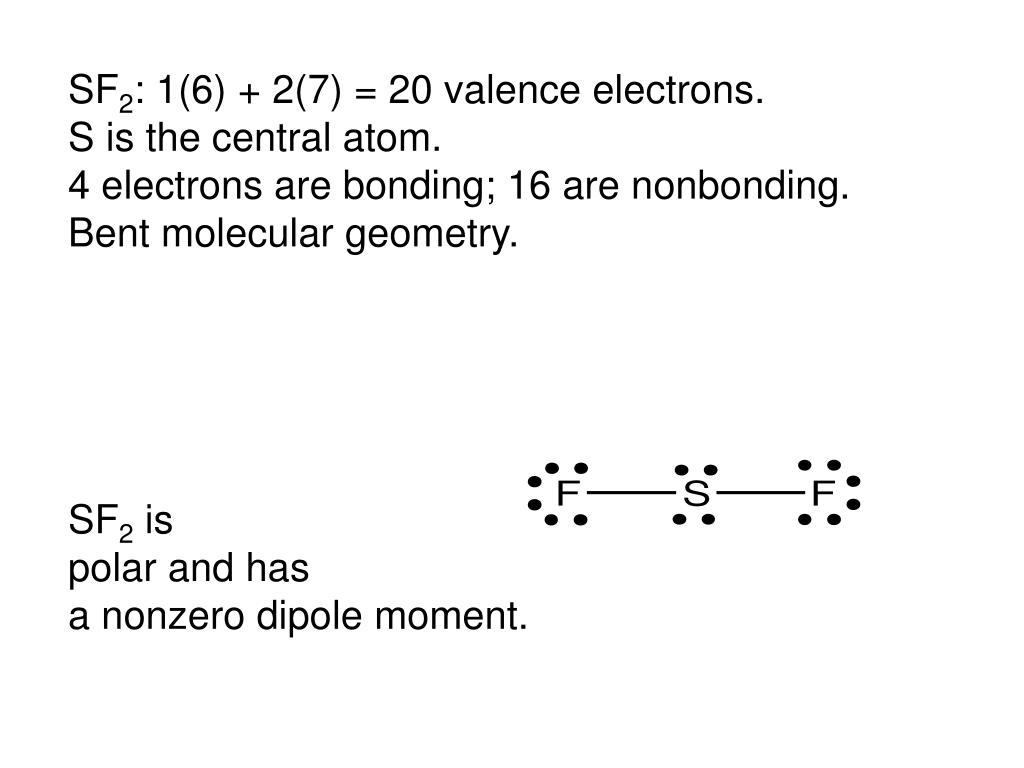
Sulfur difluoride (SF 2) is one sulfur atom connected to two fluorine atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
